How to Install MedDRA with Pinnacle 21 Community
MedDRA Dictionary
Medical Dictionary for Regulatory Activities (MedDRA) is used for coding Adverse Events and helps facilitate safety and regulatory review of clinical data. For more information, please see section 6.3.1 of the Technical Conformance Guide.
Since MedDRA is only available through a commercial subscription, it’s not included as part of Pinnacle 21 Community Validator distribution and must be provided by you or your company.
When provided, the Pinnacle 21 Validator uses five files (listed below) from the MedDRA dictionary to perform coding compliance checks. The MedDRA dictionary includes more files but only the ones listed below are needed for validation.
- pt.asc
- soc.asc
- llt.asc
- hlt.asc
- hlgt.asc
Steps to Install
We have created this easy process for you to add your company’s licensed copy of MedDRA to Pinnacle 21 Community Validator.
- From the validation screen, click "Install now" next to MedDRA.
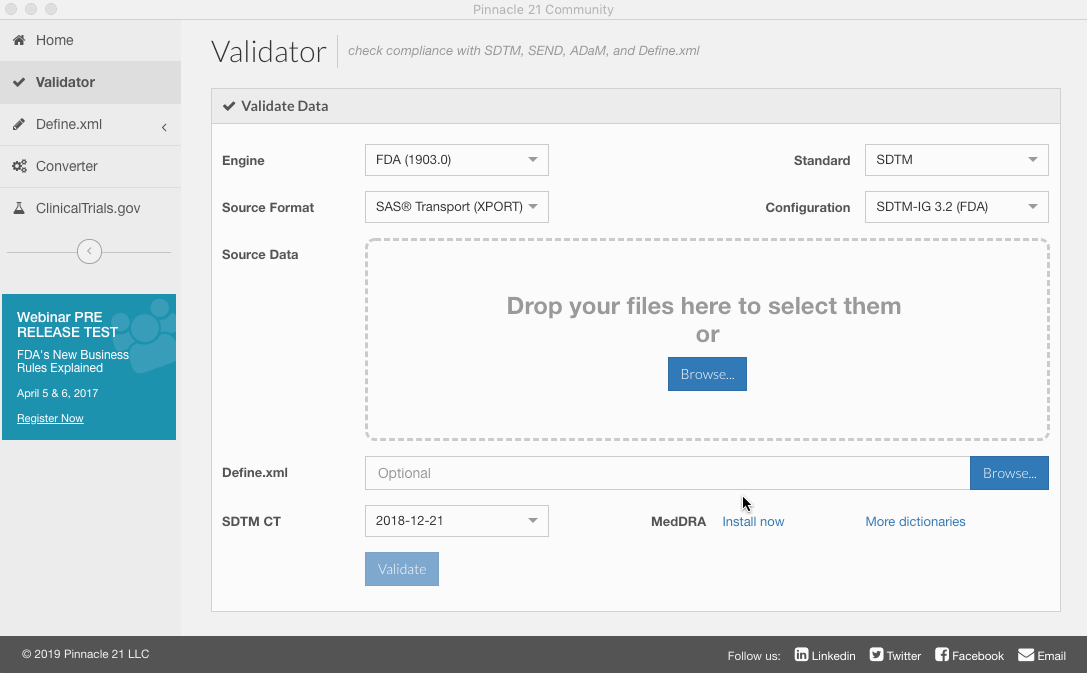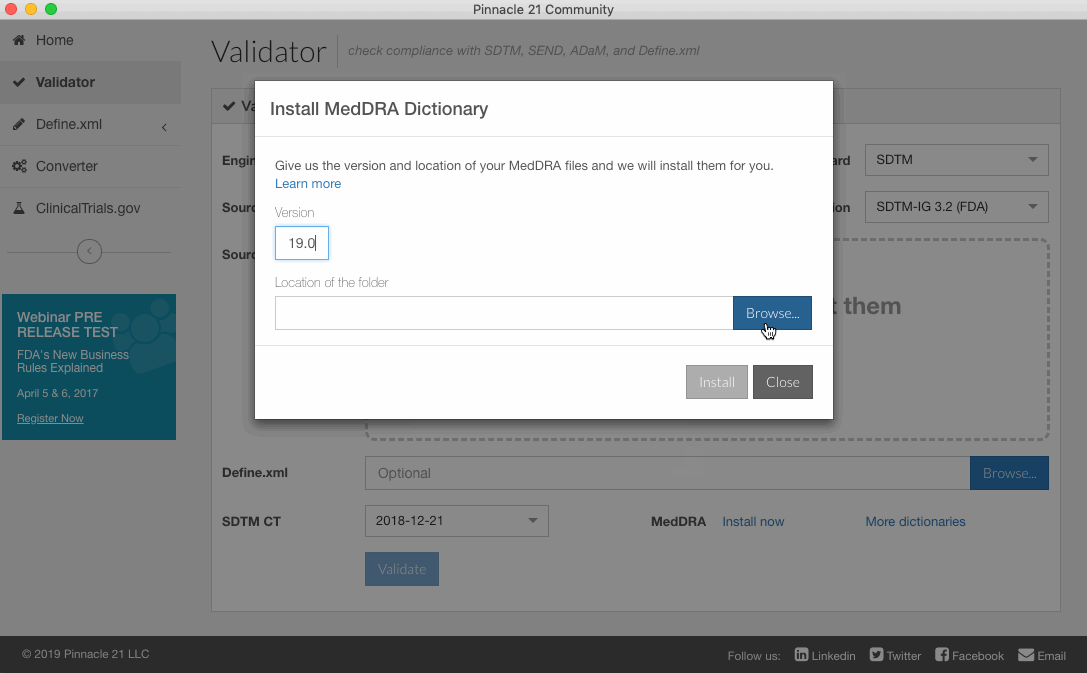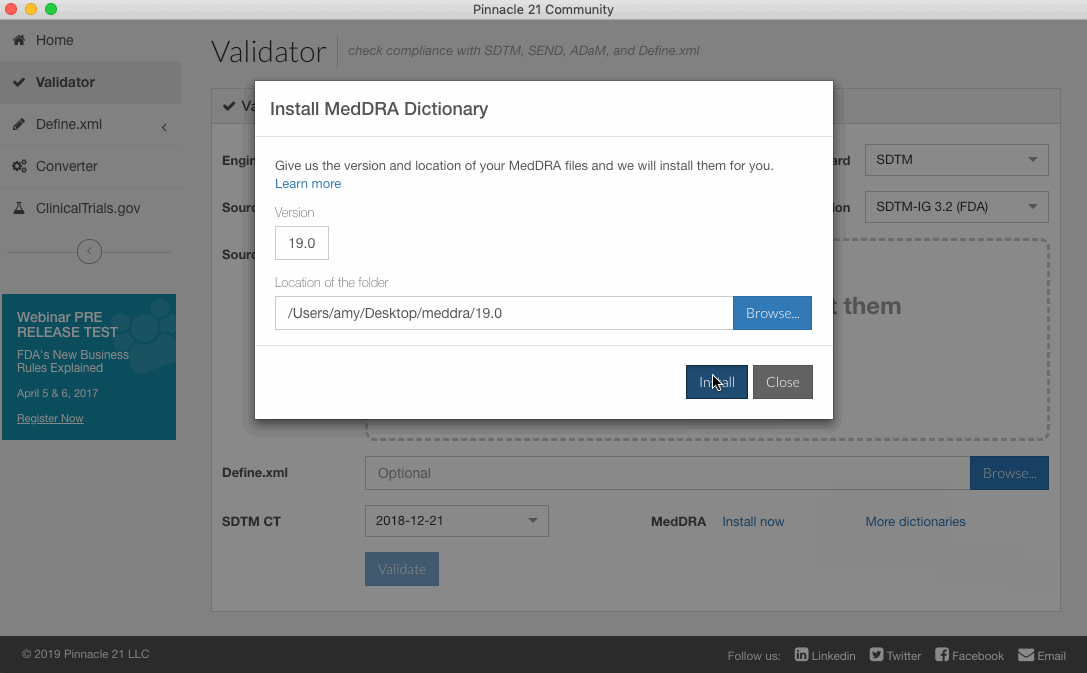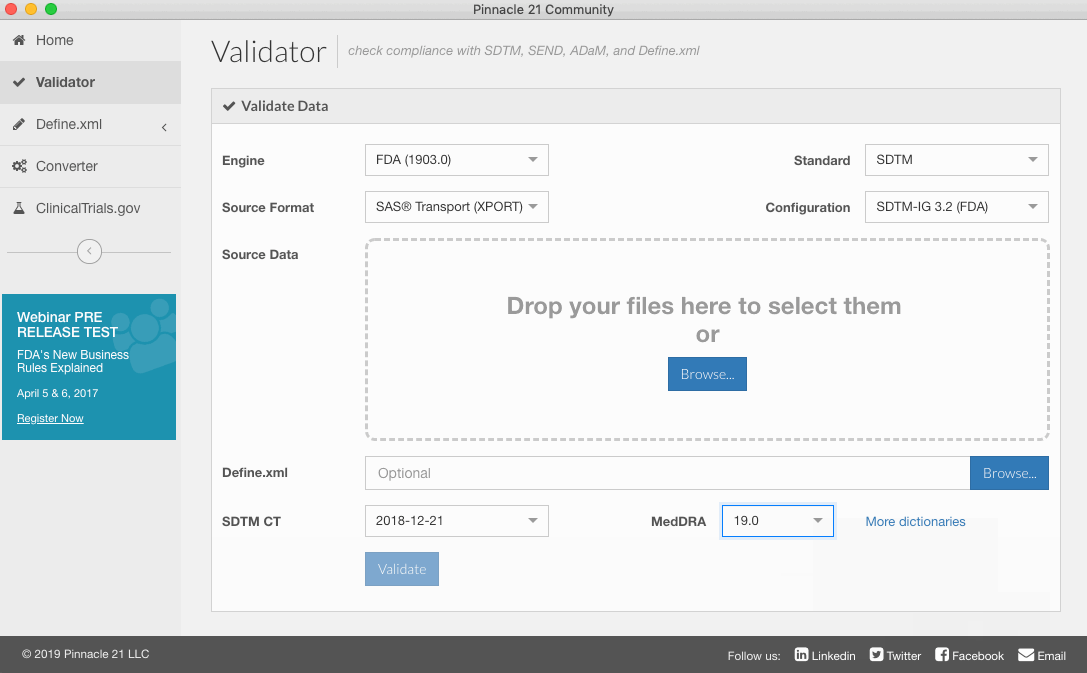
- Enter in which MedDRA version you would like to install (e.g. 20.1).
- Browse and select the the folder you would like to install. Note: the folder must contain the five files mentioned above; additional MedDRA files will be ignored.
- Click Install.
- After install is completed, click close.
After you have installed MedDRA, you will be able to select which version to use from the dropdown.
To add additional versions of MedDRA dictionary, click "More dictionaries" on the validation screen and then select the plus icon next to the dictionary. Repeat steps 2 - 5
Note: to overwrite an existing version of MedDRA already imported, follow the same steps and select "yes" when prompted if you wish to overwrite.
For any additional questions, please post on the P21 Community Forum. If you are an Enterprise client, please contact your CSM or email success@pinnacle21.com





