Define-XML 2.1 Support
About Define-XML 2.1
CDISC published version 2.1 of the Define-XML in May of 2019. It is currently only listed in the FDA data standards catalog with support beginning March 15, 2021 and becoming a requirement on March 15, 2023. In anticipation of these dates, we added support for Define-XML 2.1 starting with P21 Community 4.0.
Key Changes
- Standards can be defined at the dataset level as opposed to a single standard referenced. The define.xml document specifies which CDISC standard each dataset comes from.
- Controlled terminology can be defined at the codelist level. Some codelists may be from ADaM-CT, others from SDTM-CT, with others as non-standard.
- The Class concept was re-implemented to support the addition of SubClass.
- Representation of origin was enhanced to identify additional details of the metadata source.
How to Upgrade
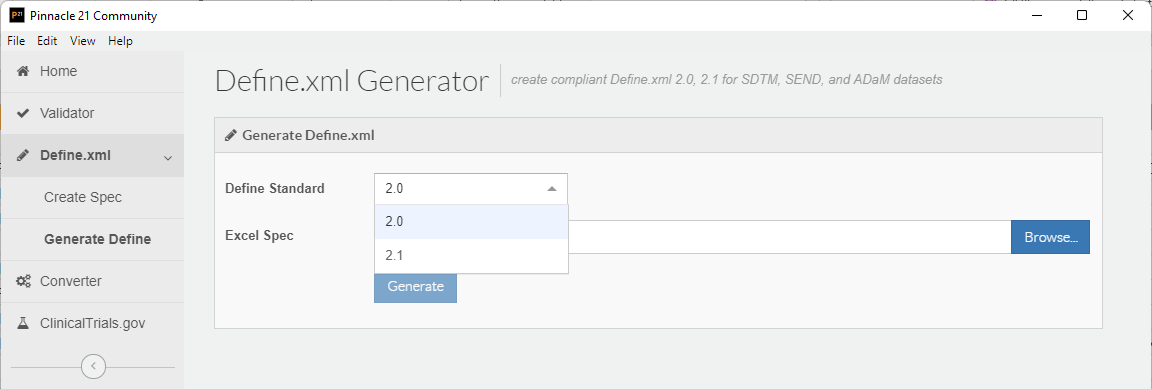
Starting with P21 Community 4.0, creating a spec in the Define.xml Generator uses a new Excel template. This new template highlights all new fields required for Define-XML 2.1 support in a peach color. Populate these new columns, then use P21 Community to generate a define.xml using the Excel spec.
Impact to Validation
Because Define-XML 2.1 allows linking of standards to datasets and controlled terminologies to codelists, it's important to make sure these values are correct. Pinnacle 21 Community uses this metadata to validate the dataset or codelist against these linked standards.
Dataset Standards
Use values that match the Define-CT codelist Standard Name (STDNAM) (C170452):
- ADaMIG
- SDTMIG
- SDTMIG-AP
- SDTMIG-MD
- SENDIG
- SENDIG-AR
- SENDIG-DART
Add the version of the standard after the name. For example: SDTMIG 3.3 or ADaMIG 1.1. Using a value other than these will produce incomplete validation results since the standard won't be checked correctly.
Codelist Terminologies
Use the base standard name (SDTM or ADaM) with a date in ISO 8601 format. For example: SDTM 2021-09-24.





