Blog
CDISC ADaM Submission Recommendations
This is the second in a series of posts where we answer questions from our recent webinar on the more commonly occurring validation rules and some potential reasons why they fired. In this post, we focus on submission recommendations.
Regulatory Expectations
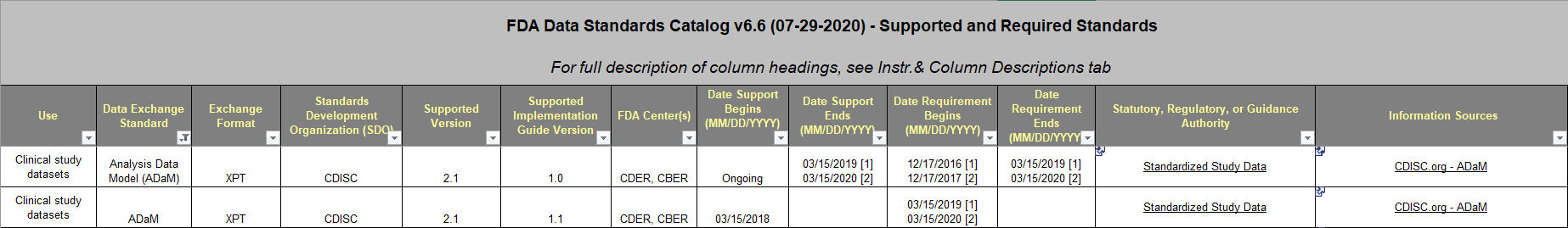
These Data Standards Catalogs from the FDA and PMDA show the valid ADaM-IG versions for your submission’s date.
- FDA currently accepts ADaM-IG 1.0 and ADaM-IG 1.1.
- Note: ADaM-IG 1.0 is only accepted for studies that began prior to the dates below. If your study started on or after these dates, then you can no longer use ADaM-IG 1.0 to submit that study.
- 03/15/2019 for NDAs, ANDAs, and certain BLAs
- 03/15/2020 for certain INDs
- Note: ADaM-IG 1.0 is only accepted for studies that began prior to the dates below. If your study started on or after these dates, then you can no longer use ADaM-IG 1.0 to submit that study.
- PMDA currently accepts ADaM-IG 1.0 only. Note: Unlike the FDA, the PMDA does not grant exceptions for issues in the Reject category.
- China's NMPA accepts CDISC datasets as of October 1st, 2020, and we’re adding more Chinese-language support across our software. To learn about implications for ADaM and SDTM datasets submitted to the NMPA, please see our recent blog post.
Using the Data Standards Catalog is easy. Navigate to the Submission Data Exchange Standards worksheet pictured below and look for the rows that are applicable to CDISC ADaM. Note: the dates in these columns are “Study start date.”
Analysis Data Reviewer’s Guide (ADRG)
You need to use the ADRG to document and explain the items below. Future releases of Enterprise will include our recommended "Fix Tips" and examples of common issue explanations.
- False positives
- Cases where you receive Vendor data that you cannot change
- Data with wrong variable labels due to different IG versions E.g., your study was started using ADaM-IG v1.0 variable labels, so the rule fired for variable label mismatch.
We recommend using the latest versions of Controlled Terminology (CT). Else you’ll need more explanations.
- E.g., SDTM’s 2018-06-29 CT does not have "Unknown" in the RACE codelist. You’d be forced to explain: “’Unknown’ was added by the sponsor to the extensible CDISC codelist ‘RACE.’”
The P21 Issues Report spreadsheet is not a required part of the submission package for the FDA or PMDA, but is helpful to include. ADRG Section 6 is sufficient to meet the requirement. More to Come We hope these points assist you in your workflow. Please check back as we continue this series of posts!
More to Come
We hope these points assist you in your workflow. Please check back as we continue this series of posts!
Tags:
Blog Main Page